Lexapro and Nutrient Depletions
What Your Doctor Didn't Tell You
If you are taking Lexapro, or considering it, there is a good chance nobody has told you about this. Not because your doctor doesn't care. But because the standard prescribing visit does not include a conversation about what happens to your nutrient status, your liver enzymes, or your body's ability to make serotonin in the first place when you start this medication.
This post is not anti-medication. Lexapro helps a lot of people. But informed use means understanding the full picture, including what your body needs to support the medication, what can interfere with it, and what questions are worth asking your provider.
If you want the deep clinical version, watch the lecture embedded below. If you want the plain-language breakdown, keep reading.
If you would like a copy of the slides, contact me here.
What Is Lexapro and What Is It Approved For?
Lexapro is the brand name for escitalopram oxalate. It is a Selective Serotonin Reuptake Inhibitor, or SSRI, which is a class of medications that work in the brain to regulate mood.
The FDA approved Lexapro in 2002 for two specific conditions:
Major Depressive Disorder (MDD) in adults and adolescents aged 12 to 17
Generalized Anxiety Disorder (GAD) in adults only
It is not FDA approved for children under 12 for depression, and it is not approved for anxiety in anyone under 18. The standard starting dose is 10mg once daily, with a maximum of 20mg. Research showed no additional benefit at the higher dose for most people (FDA Prescribing Information, 2017).
How Does Lexapro Work?
Here is the simple version. Serotonin is a brain chemical that helps regulate mood, sleep, and emotional response. Normally, after serotonin is released in the brain, it gets pulled back into the nerve cell that released it. Lexapro blocks that process, called reuptake, so serotonin stays in the space between nerve cells longer.
This increases serotonin activity in the brain. The medication starts affecting serotonin almost immediately, but mood improvement typically takes one to four weeks.
The reason for the delay is that the brain needs time to adapt and reorganize around the change. Some early symptom improvement can appear in the first week, but full benefit requires neuroadaptive changes over time (Taylor et al., 2006; Briley & Moret, 1993).
Here is what most people are never told: Lexapro works on the serotonin your brain already has. But your brain needs specific nutrients to make serotonin in the first place. If those nutrients are low, the medication may have less to work with.
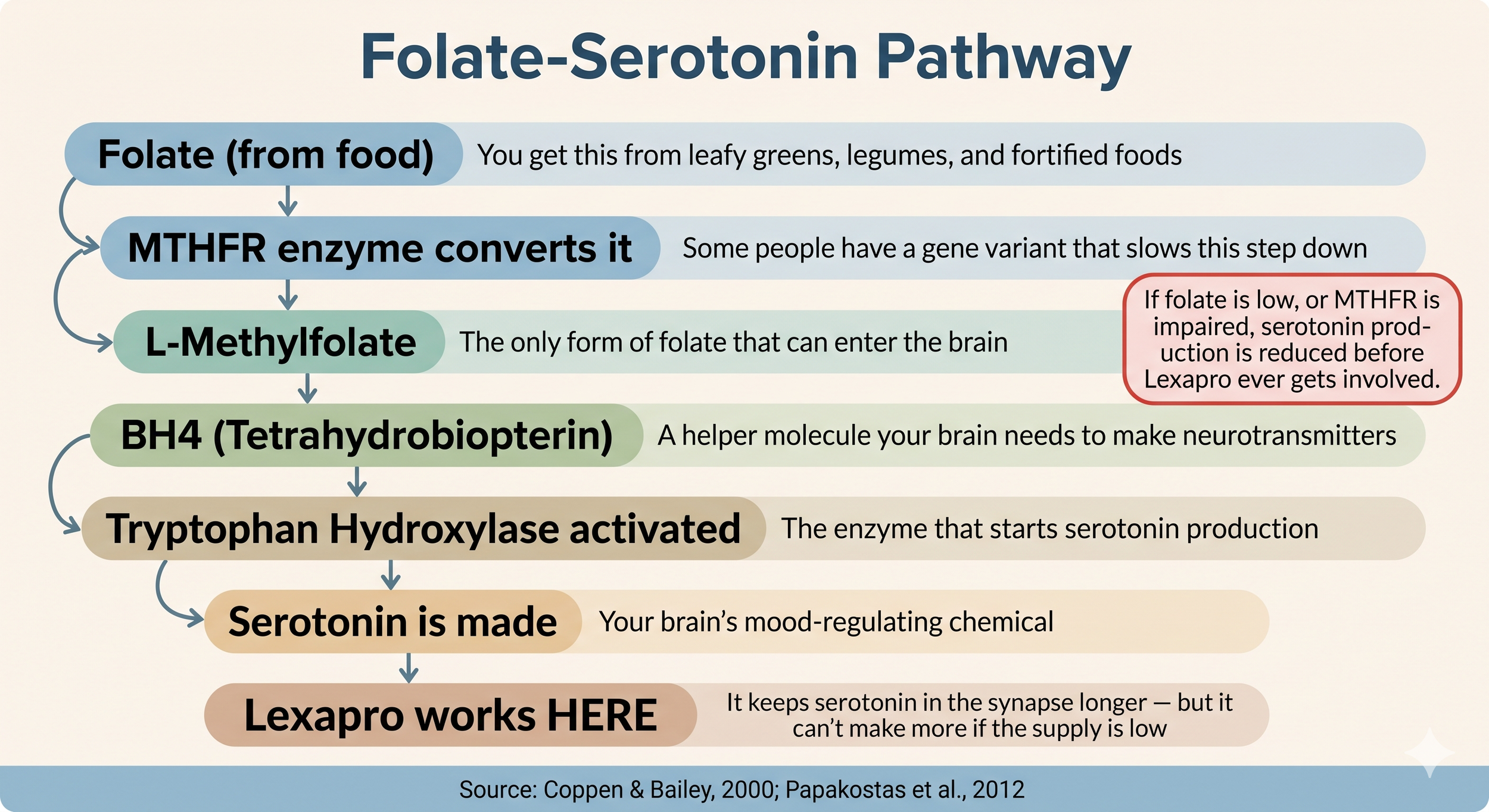
The Folate-Serotonin Paradox: The Part That Changes Everything
This is the part of the conversation that almost never happens in a standard prescribing visit. Your brain needs a specific form of folate called L-methylfolate to produce serotonin. L-methylfolate is the only form of folate that can cross the blood-brain barrier and get into the brain where it is needed. Once there, it helps produce a molecule called BH4, which activates the enzyme that starts serotonin production. No L-methylfolate, reduced BH4, less serotonin being made (Papakostas et al., 2012).
Lexapro tries to optimize the serotonin your brain has. But if your folate status is low, or if a genetic variant called MTHFR is impairing your ability to convert folate into its active form, your brain is not making enough serotonin for the medication to work with in the first place.
The research supports this. A randomized placebo-controlled trial by Coppen and Bailey (2000) found that women on fluoxetine, a similar SSRI, plus folic acid had a 93.9% response rate compared to 61.1% in the group taking fluoxetine alone. The only variable was folate. A separate study by Fava et al. (1997) found that patients with low folate were significantly less likely to respond to antidepressant treatment and more likely to have a severe form of depression.
A more recent set of clinical trials found that adding L-methylfolate 15mg per day to an ongoing SSRI produced a 32% response rate in people who had not responded to SSRIs alone, compared to 15% in the placebo group (Papakostas et al., 2012).
It is worth noting that most of this research uses fluoxetine rather than escitalopram specifically. Research on individual SSRIs and folate is limited. But the folate-BH4-serotonin mechanism is upstream of where all SSRIs work, so the finding applies to the drug class as a whole.
For people with ADHD, PMDD, or neurodivergent kids, this is especially relevant. Research shows this population has higher rates of MTHFR variants that impair folate conversion. This does not mean everyone with MTHFR will have a poor response to Lexapro. It means folate status is worth knowing.
The FDA Warnings That Are Rarely Discussed With Patients
The Lexapro prescribing information contains warnings that most patients are never walked through at the time of prescribing. These are not rare edge cases. They are documented, clinically significant risks that deserve a conversation.
Black box warning on suicidality
Lexapro and other antidepressants carry a black box warning for increased risk of suicidal thinking in children, adolescents, and young adults under 24, especially in the first weeks of treatment or at dose changes. Patients and families should be told to watch for new or worsening mood changes, agitation, or unusual behavior and report them promptly (FDA Prescribing Information, 2017).
Serotonin syndrome
Serotonin syndrome is a potentially life-threatening reaction that can occur when too much serotonin builds up in the body. It is most likely when Lexapro is combined with other serotonergic medications or certain supplements. Symptoms include agitation, rapid heart rate, high blood pressure, sweating, muscle twitching, and in severe cases, loss of coordination or confusion. St. John's Wort, triptans, tramadol, and certain antidepressants are among the most common triggers when combined with Lexapro (FDA Prescribing Information, 2017; Edinoff et al., 2021).
Discontinuation syndrome
Stopping Lexapro suddenly can cause discontinuation syndrome. Symptoms include electric shock-like sensations, dizziness, mood swings, irritability, nausea, and anxiety. These are not signs of addiction. They are the nervous system adjusting to a rapid change. A gradual taper is always recommended. Because escitalopram has a shorter half-life than some other SSRIs, stopping it abruptly is more likely to cause noticeable withdrawal symptoms (FDA Prescribing Information, 2017; Strawn et al., 2023).
Hyponatremia (low sodium)
SSRIs, including escitalopram, carry the highest risk of hyponatremia (low sodium) among all antidepressants. Escitalopram was specifically named as a higher-risk agent in research examining sodium and antidepressants (Wilkinson et al., 1999). Symptoms include headache, difficulty concentrating, memory changes, confusion, weakness, and in severe cases, seizure. Women, older adults, and those on diuretics are at greatest risk. Sodium monitoring early in treatment is clinically reasonable.
Bleeding risk
Lexapro affects how platelets function, which can increase the risk of bleeding. This risk is compounded when combined with NSAIDs like ibuprofen, aspirin, or blood thinners. Ginkgo biloba, which has its own blood-thinning properties, can add to this risk as well (FDA Prescribing Information, 2017).
Activation syndrome in children and adolescents
In youth, SSRIs can cause a cluster of symptoms called activation syndrome: increased restlessness, irritability, impulsivity, insomnia, and anxiety. These symptoms overlap with the disorders being treated, so they are often missed or misattributed to worsening of the original condition. Activation tends to emerge early in treatment or after dose increases (Strawn et al., 2023).
Common Adverse Reactions and What the Research Shows Beyond the Label
The FDA lists the following as the most common adverse reactions occurring in 5% or more of patients at twice the rate of placebo:
Insomnia
Nausea
Fatigue and somnolence
Increased sweating
Decreased libido
Ejaculation disorder
Anorgasmia
The research literature shows the picture is broader than this. A 2021 narrative review identified 86 case reports connecting SSRIs including escitalopram to extrapyramidal symptoms, which are movement-related side effects including restlessness, muscle stiffness, and involuntary movements. Most occurred within 30 days of starting or increasing the dose (Edinoff et al., 2021).
Additional adverse effects documented in the research include QT prolongation (a heart rhythm change), skin reactions including photosensitivity and rarely Stevens-Johnson syndrome, weight and body composition changes, and a possible association with cataracts on long-term use. Sexual dysfunction is widely underreported and contributes meaningfully to people stopping the medication (Edinoff et al., 2021; Strawn et al., 2023).
None of this means Lexapro is dangerous. It means understanding the full profile helps patients and providers make better decisions together.
Nutrient Status and SSRI Users: What the Data Shows
A 2018 study using National Health and Nutrition Examination Survey (NHANES) data looked at 655 adults who had been prescribed an SSRI for at least 180 days and compared them to 12,372 non-users. The findings were significant.
Among SSRI users:
SSRI use was associated with a measurable reduction in bone mineral density
Over half had inadequate daily intake of calcium, vitamin D, and potassium
Inadequate vitamin D intake was associated with lower bone density in both SSRI users and non-users
The combination of SSRI use and inadequate zinc intake was specifically associated with low bone density (Kindilien et al., 2018)
To be clear: this research does not prove that Lexapro directly depletes these nutrients. What it shows is that people taking SSRIs as a population are commonly low in key nutrients, and that those deficiencies affect measurable health outcomes. The authors of the study specifically recommended nutritional counseling and micronutrient monitoring for patients prescribed SSRIs.
Monitoring and understanding nutrient status is not about adding more supplements. It is about knowing what the baseline looks like so informed decisions can be made.
Drug and Nutrient Interactions With Lexapro
Escitalopram is metabolized by CYP3A4 and CYP2C19 liver enzymes and is a weak inhibitor of CYP2D6. This means several medications and substances can affect how much Lexapro stays in the body and how it behaves.
Cannabis (THC and CBD)
Co-administration of escitalopram with THC or CBD has been shown to significantly increase the half-life and concentration of escitalopram in the body by inhibiting cytochrome enzyme activity. In plain terms, cannabis can cause the medication to stay in the system longer and at higher levels than intended (Jung et al., 2024).
Ashwagandha (Withania somnifera)
Combining escitalopram with ashwagandha has been reported to cause myalgia, epigastric pain, nausea, vomiting, restless legs syndrome, and severe cough. The interaction is thought to involve CYP3A4 and CYP2D6 pathways (Jung et al., 2024).
Rhodiola rosea
Interactions between escitalopram and Rhodiola rosea have been reported including cough, ejaculation disorders, bleeding, and ventricular arrhythmia (Jung et al., 2024).
NSAIDs, aspirin, and blood thinners
This combination is listed directly in the FDA prescribing information. Lexapro affects platelet function and serotonin release from platelets. Adding drugs that affect clotting compounds this risk and increases the likelihood of gastrointestinal or other bleeding (FDA Prescribing Information, 2017).
MAO inhibitors
MAO inhibitors are contraindicated with Lexapro. A minimum 14-day washout is required before starting escitalopram after stopping an MAOI. Combining them can cause serious or life-threatening serotonin syndrome (FDA Prescribing Information, 2017).
Herbs to Discuss With Your Provider Before Taking Lexapro
Herbal medicine is powerful. That is exactly why it deserves the same careful consideration as prescription medications when it comes to interactions.
St. John's Wort
This is the most important herb caution with Lexapro. St. John's Wort is listed directly in the FDA prescribing information as a known serotonin syndrome risk. Do not combine with Lexapro. The risk is not theoretical (FDA Prescribing Information, 2017; Zuo et al., 2022).
Kava
Kava may affect the liver enzymes that break down Lexapro, potentially changing how much medication stays in your system. Kava also carries its own independent liver safety concerns regardless of other medications being used (Zuo et al., 2022).
Black Cohosh
Black cohosh is commonly used for hormonal and perimenopausal symptoms. In vitro studies show it may affect CYP2D6, CYP2C19, and CYP3A4, though clinical trial results are mixed. Until more consistent data exists, disclosing use to your provider is the right call (Zuo et al., 2022).
Ginkgo Biloba
Ginkgo has blood-thinning properties of its own. Because Lexapro already increases bleeding risk, combining the two may compound that effect. Discuss with your provider before using both (Zuo et al., 2022).
Supportive Strategies That Make Sense Clinically
These strategies are not replacements for medication. They are context for medication. The goal is to support the foundation the medication is working on.
Folate status
Given the research on folate and antidepressant response, folate status is worth knowing. L-methylfolate is the active form that crosses the blood-brain barrier. For people with known or suspected MTHFR variants, standard folic acid may not convert efficiently. Testing before supplementing is always preferred over guessing.
Vitamin D
Over half of SSRI users in the Kindilien et al. (2018) study had inadequate vitamin D intake. Vitamin D supports mood, immune function, and bone health. Sunlight exposure and dietary sources are preferred first. Testing is the most accurate way to know where you stand.
Zinc
Inadequate zinc combined with SSRI use was specifically associated with lower bone density in the NHANES study. Zinc also plays a role in neurotransmitter function. It is a commonly deficient nutrient in the neurodivergent population.
Omega-3 fatty acids
Whole food sources such as fatty fish and eggs are preferred first. Omega-3s may support neurological and mood function and are broadly supportive for the nervous system.
Sleep consistency
Consistent sleep and wake timing supports serotonin regulation. Insomnia is one of the most common side effects of Lexapro, so addressing sleep hygiene is clinically relevant and not just lifestyle advice.
Sodium monitoring
Particularly relevant for women, older adults, and those on diuretics. Electrolyte monitoring early in antidepressant treatment is clinically reasonable given the documented hyponatremia risk specific to escitalopram (Wilkinson et al., 1999).
Lexapro is not inherently good or bad. It is powerful.
Powerful tools work best when the foundation is solid. If you are on Lexapro and still not feeling the way you expected, or if you are experiencing side effects that were never explained to you, the answer may not be a higher dose or a different medication. It may be what is happening in the body underneath the prescription.
Informed care is safer care.
Want to Understand Your Labs Before Making Any Changes?
My Read Between the Labs session was made for exactly this. For $99 I will review your existing blood work, walk you through what I am seeing alongside your medications and supplements, and give you a written summary within 72 hours. No guessing. No overwhelm.
References
Briley, M., & Moret, C. (1993). Neurobiological mechanisms involved in antidepressant therapies. Clinical Neuropharmacology, 16(5), 387-400.
Coppen, A., & Bailey, J. (2000). Enhancement of the antidepressant action of fluoxetine by folic acid: A randomised, placebo controlled trial. Journal of Affective Disorders, 60(2), 121-130. https://doi.org/10.1016/S0165-0327(00)00153-1
Edinoff, A. N., Akuly, H. A., Hanna, T. A., Ochoa, C. O., Patti, S. J., Ghaffar, Y. A., Kaye, A. D., Viswanath, O., Urits, I., Boyer, A. G., Cornett, E. M., & Kaye, A. M. (2021). Selective serotonin reuptake inhibitors and adverse effects: A narrative review. Neurology International, 13(3), 387-401. https://doi.org/10.3390/neurolint13030038
Fava, M., Borus, J. S., Alpert, J. E., Nierenberg, A. A., Rosenbaum, J. F., & Bottiglieri, T. (1997). Folate, vitamin B12, and homocysteine in major depressive disorder. The American Journal of Psychiatry, 154(3), 426-428.
Jung, Y. S., Jin, B. H., Choi, J. E., Park, M. S., Kim, Y. W., Kang, H. W., Cho, S., & Kim, C. O. (2024). Assessment of pharmacokinetic effects of herbal medicines on escitalopram. Therapeutics and Clinical Risk Management, 20, 151-160. https://doi.org/10.2147/TCRM.S448090
Kindilien, S., Goldberg, E. M., Roberts, M. H., & Gonzales-Pacheco, D. (2018). Nutrition status, bone mass density, and selective serotonin reuptake inhibitors. Preventive Medicine, 113, 62-67. https://doi.org/10.1016/j.ypmed.2018.05.008
Lanzenberger, R. (2011). The molecular and functional network in SSRI treatment. European Psychiatry, 26(S2), 2222-2222. https://doi.org/10.1016/S0924-9338(11)73924-4
Papakostas, G. I., Shelton, R. C., Zajecka, J. M., Etemad, B., Rickels, K., Clain, A., Baer, L., Dalton, E. D., Carter-Campbell, J. T., Rosenbaum, J. F., Har-Noy, B. S., Mischoulon, D., Bottiglieri, T., Cassiello, C. F., Swee, M. B., & Fava, M. (2012). L-methylfolate as adjunctive therapy for SSRI-resistant major depression. American Journal of Psychiatry, 169(12), 1267-1274. https://doi.org/10.1176/appi.ajp.2012.11071114
Strawn, J. R., Mills, J. A., Poweleit, E. A., Ramsey, L. B., & Croarkin, P. E. (2023). Adverse effects of antidepressant medications and their management in children and adolescents. Pharmacotherapy, 43(7), 675-690. https://doi.org/10.1002/phar.2767
Taylor, M. J., Freemantle, N., Geddes, J. R., & Bhagwagar, Z. (2006). Early onset of selective serotonin reuptake inhibitor antidepressant action. Archives of General Psychiatry, 63(11), 1217-1223. https://doi.org/10.1001/archpsyc.63.11.1217
U.S. Food and Drug Administration. (2017). Lexapro (escitalopram oxalate) prescribing information. Allergan USA, Inc.
Wilkinson, T. J., Begg, E. J., Winter, A. C., & Sainsbury, R. (1999). Incidence and risk factors for hyponatraemia following treatment with fluoxetine or paroxetine in elderly people. British Journal of Clinical Pharmacology, 47(2), 211-217. https://doi.org/10.1046/j.1365-2125.1999.00872.x
Woron, J., & Siwek, M. (2018). Unwanted effects of psychotropic drug interactions with medicinal products and diet supplements containing plant extracts. Psychiatria Polska, 52(6), 983-996. https://doi.org/10.12740/PP/OnlineFirst/80998
Zuo, H.-L., Huang, H.-Y., Lin, Y.-C.-D., Cai, X.-X., Kong, X.-J., Luo, D.-L., Zhou, Y.-H., & Huang, H.-D. (2022). Enzyme activity of natural products on cytochrome P450. Molecules, 27(2), 515. https://doi.org/10.3390/molecules27020515